 Study with Quizlet and memorize flashcards containing terms like Aristotles theory, Aristotle Model, Democritus Picture and more. I started this with my students (year 10) in class, then had them finish it as homework, but it could be used completely as homework or completely as a class activity (especially with younger students), reading out the information sentence-by-sentence and building up the timeline together. Schrodinger and Other Scientists Timeline date of theory. 1935 Niels Bohr presents his analysis of the EPR paradox. 1935 Henry Eyring develops the transition state theory. 1935 Albert Einstein, Boris Podolsky, and Nathan Rosen put forth the EPR paradox. The four examples are also included here as. 1935 Hideki Yukawa presents a theory of the nuclear force and predicts the scalar meson. Dalton thought that atoms were the smallest units of matter tiny, hard spheres that could not be broken down any further. However, one of his underlying assumptions was later shown to be incorrect. The fourth and fifth pages have pictures of some of my students' timelines. Daltons ideas proved foundational to modern atomic theory. I instructed my pupils, rather than printing this page out. 1913 - Bohr Bohr proposed the theory that the electrons orbit the nucleus in a predetermined route. 
His model postulated that atoms consist of a big positively-charged sphere studded with negatively charged electrons (he called them 'corpuscles') like fruit in a plum pudding. The fourth page has instructions on how to set up a book for the timeline (as a double page wasn't enough). Thomson proposed the 'plum pudding' theory of the divisible atom in 1904, after discovering electrons in 1897. 4) A chemical reaction is a rearrangement of atoms. Focuses on discoveries required for AQA additional physics (P2.5.1 atomic structure), including plum pudding model, Rutherfords gold foil experiment, and the subsequent Rutherford and Bohr models. 2) All atoms of a given element are identical in mass and properties 3) Compounds are formed by a combination of two or more different kinds of atoms. Activity for students to learn (or revise) the evolution of the atom. Atoms are indivisible and indestructible. 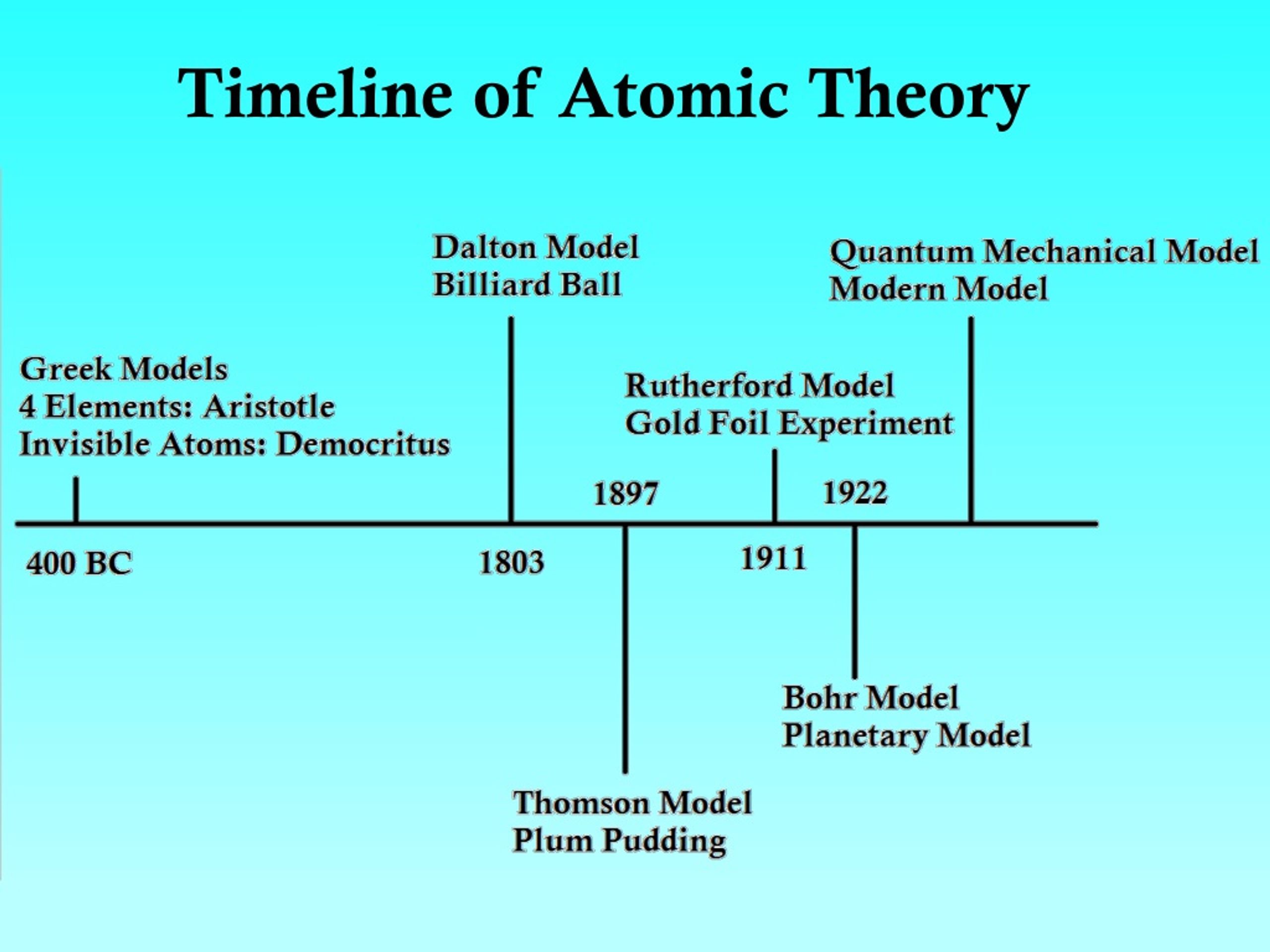
The third page has pictures of all of the scientists (and philosopher) named in the history (print 1 page for 2 pupils). Dalton’s Atomic Theory 1803 1) All matter is made of atoms. 
The first two pages of the word document is a summary of how the atomic model has changed with diagrams of the models. Focuses on discoveries required for AQA additional physics (P2.5.1 atomic structure), including plum pudding model, Rutherford's gold foil experiment, and the subsequent Rutherford and Bohr models. In 1913, Niels Bohr published a groundbreaking paper that introduced a new way of understanding atomic phenomena (). Attempts to trace precisely how Dalton developed this theory have proved futile even Dalton’s own recollections on the subject are incomplete. AND if they already knew that the electron was small and negative, then the atom must have a small positive nucleus with the electrons around them.Activity for students to learn (or revise) the evolution of the atom. John Dalton - Atomic Theory, Chemistry, Physics: By far Dalton’s most influential work in chemistry was his atomic theory. Atoms of different elements are different from each other and can be. Atoms of the same element are identical with respect to their weights. If the positive alpha particles mostly passed through the foil, but some bounced back. In1803 after many years of researching atoms John Dalton published his Theory on atoms which stated All elements are made up of tiny indivisible particles, known as atoms. Daltons Atomic Theory (1803) British chemist John Dalton proposed that matter is composed of indivisible, indestructible tiny particlesatoms. How could that be if the plumb pudding model was correct? Rutherford's experiment prompted a change in the atomic model. When talking about the atomic model timeline project, the Scientists understanding of atoms has gone through several stages, and the following are five main historical models. Rutherford found that most of them went right through the foil. Study the atomic theory timeline, how scientists modeled an atom including discoveries of parts of the atom, and what an atom is today. If you shoot these positive alpha particles at this positive pudding atom, they should mostly bounce off, right? Well, that is not what happened. He shot some alpha particles (which are really just the nucleus of a helium atom) at some really thin gold foil. 1704 Isaac Newton theorized a mechanical universe with small, solid masses in motion. Ernest Rutherford said one day "hey, I think I will shoot some stuff at atoms." I am sure his wife said "oh, Ernie" (she probably called him Ernie) "if it makes you happy to play with your little physics stuff, go ahead. Democritus’ atomic theory posited that all matter is made up small indestructible units he called atoms. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
